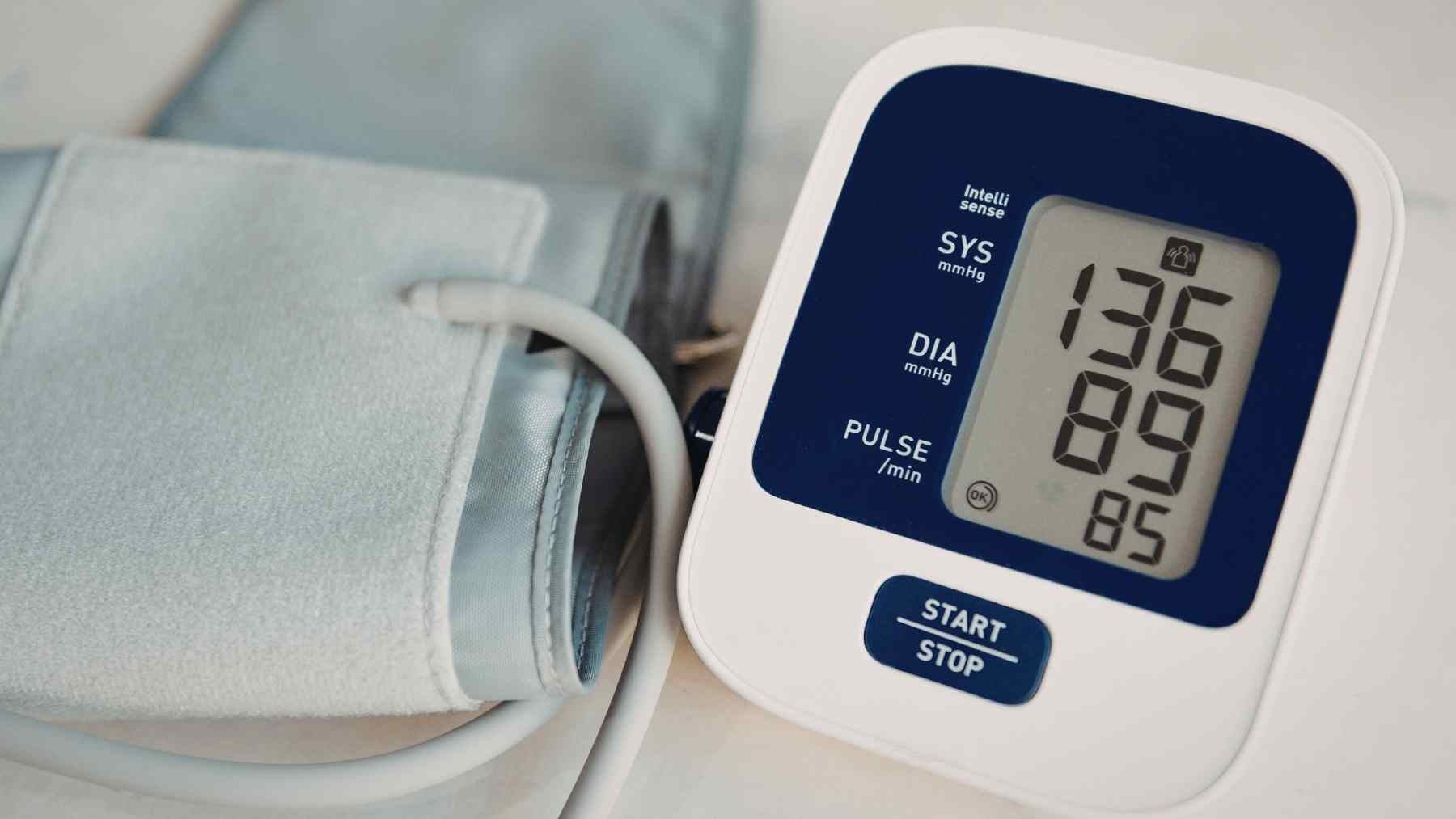
A stubborn blood pressure reading can feel like a private frustration, especially when a person is already taking medication and doing the right things. Now, an experimental pill called baxdrostat is drawing attention because it appears to lower hard-to-control blood pressure in people with chronic kidney disease, a group that often has few easy options.
The bigger story is not just the number on the cuff. In a Phase 2 trial, baxdrostat also reduced albumin in the urine, a warning sign linked to kidney and cardiovascular risk, raising hope that the drug may one day help slow the dangerous cycle between high blood pressure and kidney damage.
A dangerous cycle
High blood pressure and chronic kidney disease often push each other in the wrong direction. High pressure can damage the tiny blood vessels in the kidneys, and weaker kidneys can make blood pressure even harder to control.
The CDC estimates that about 14% of U.S. adults, or 37 million people, have chronic kidney disease. It also reports that more than 2 in 10 adults with high blood pressure are estimated to have CKD, which shows why this trial matters beyond the lab.
How baxdrostat works
Baxdrostat targets aldosterone, a hormone made by the adrenal glands. Aldosterone helps the body manage salt and water, but when it is too active, the body may retain more sodium and fluid, pushing blood pressure higher.
In practical terms, that means the drug is trying to quiet one of the systems that keeps pressure elevated even after standard treatment. The American Heart Association said baxdrostat belongs to a class of medicines that inhibit aldosterone production, but it is still being studied and is not yet a routine kidney treatment.
What the trial found
The study included 195 people, and 192 were randomly assigned to receive baxdrostat or placebo in addition to standard care. Their average starting systolic blood pressure was about 151 mm Hg, even though they were already taking an ACE inhibitor or an ARB.
After 26 weeks, systolic blood pressure fell by an average of 8.1 mm Hg more in the baxdrostat groups than in the placebo group. That may sound small at the kitchen table, but in blood pressure medicine, even single-digit reductions can be meaningful for heart, stroke, and kidney risk.
The kidney signal
The most eye-catching kidney finding involved albumin, a protein that should not usually spill heavily into urine. High urine albumin can signal kidney stress and a higher risk of future cardiovascular problems.
In an exploratory analysis, urine albumin levels were 55% lower among participants taking baxdrostat than among those taking placebo. Lead author Jamie P. Dwyer, M.D., said, “The reduction in urine albumin gives us hope” that the drug may help delay kidney damage, though larger studies still need to prove that.
The safety warning
There was an important caution. High potassium levels occurred in 41% of participants taking baxdrostat, compared with 5% of those taking placebo, and most cases were described as mild to moderate.
That matters because high potassium can be dangerous, especially for people with kidney disease. No deaths or unexpected adverse events were reported in the trial, but serious adverse events occurred in 9% of baxdrostat participants and 3% of placebo participants.
Why doctors are watching
People with chronic kidney disease have often been left out of drug trials, even though they are among the patients most likely to struggle with blood pressure control. That is one reason this study is getting attention from kidney and heart specialists.
Jordana B. Cohen, M.D., who was not involved in the trial, called the class a possible “game changer” for this patient group. That is a strong phrase, but the evidence still has to move from promising markers to proven long-term outcomes.
More evidence is building
Baxdrostat has also shown results in larger Phase 3 hypertension trials. In the Bax24 trial, AstraZeneca reported a placebo-adjusted reduction of 14.0 mm Hg in 24-hour average systolic blood pressure after 12 weeks in people with resistant hypertension.
The company also said the drug’s New Drug Application was accepted under FDA Priority Review for adults with hard-to-control hypertension as an add-on to other blood pressure medicines. The expected FDA action window was the second quarter of 2026, so the regulatory story is still unfolding.
What patients should know
For now, baxdrostat is not a do-it-yourself solution or a replacement for medical care. People with chronic kidney disease should keep working with their clinicians on blood pressure, blood sugar, cholesterol, salt intake, and medication safety.
Still, the findings point to a useful idea. Treating stubborn blood pressure may require looking beyond the cuff and focusing on the heart, kidneys, hormones, and daily habits as one connected system.
The study was published on the Journal of the American Society of Nephrology.